|
Creating Medical Plastics That Heal Themselves
New polymers could allow for self-healing materials, the University of
Pittsburgh recently reported.
Materials manufactured from the new composites can regenerate themselves when
damaged. Results of the Pitt research were recently published in the journal
Nano Letters by the American Chemical Society.
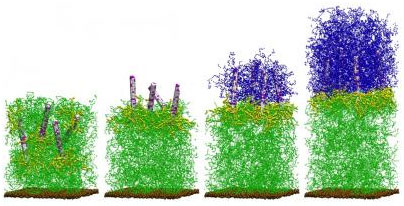
An image of the self-generating composite.
Submitted art: University of Pittsburgh
The researchers drew inspiration from animals that can regenerate missing or
severed limbs. These processes are guided by three unique instruction sets,
described by the study authors as initiation, propagation, and termination.
This threefold process, also known as a dynamic cascade, was replicated by
researchers in a synthetic material. However, developing the self-healing
composite was no easy feat. Because animals and other living organisms can
transport building materials through a circulatory system, it is relatively
simple for an organism to transport the materials it needs to a regeneration
site. However, synthetic materials don’t have such systems.
To create a sensor that initiated and controlled the regeneration process,
researchers created a hybrid material featuring nanorods embedded inside a
polymer gel. This composite is then saturated with a solution contain
cross-linkers and monomers, allowing for a synthetic replica of a biological
cascade.
Because the functionalized chains on nanorods keeps them localized at the
interface, the initiator sites along the surface of a rod can trigger the
desired polymerization process with the crosslinker and monitors in the
solution. Each of the nanorods has a diameter of 10 nanometers.
As the next step, researchers hope to improve the binding between new and older
gels. To make this possible, researchers once again looked to nature. "One
sequoia tree will have a shallow root system, but when they grow in numbers, the
root systems intertwine to provide support and contribute to their tremendous
growth," states Dr. Anna Balazs, principal investigator.
”While others have developed materials that can mend small defects, there is no
published research regarding systems that can regenerate bulk sections of a
severed material. This has a tremendous impact on sustainability because you
could potentially extend the lifetime of a material by giving it the ability to
regrow when damaged.”
A group of European researchers announced research findings on a separate
self-healing material in September.
http://www.qmed.com/news/creating-medical-plastics-heal-themselves
MedTechs Tackle Replacing 'Workhorse' Plasticizer
 The jury is still out over whether phthalate plasticizers such as DEHP really
cause health problems in humans, according to Peter M. Galland of Teknor Apex.
But publics and governments in the U.S. and Europe have become convinced enough
that medical device companies will have to find replacements for them anyway. The jury is still out over whether phthalate plasticizers such as DEHP really
cause health problems in humans, according to Peter M. Galland of Teknor Apex.
But publics and governments in the U.S. and Europe have become convinced enough
that medical device companies will have to find replacements for them anyway.
Confronting growing evidence that exposure to phthalate plasticizers could cause
a range of health issues, Congress declared in the Consumer Product Safety
Improvement Act of 2008 that children's toys and items such as pacifiers could
no longer contain di-(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP),
and benzyl butyl phthalate (BBP). At the same time, it provisionally prohibited
three other phthalates: diisononyl phthalate (DINP), diisodecyl phthalate (DIDP),
and di-n-octyl phthalate (DNOP). Then, in December 2012, FDA restricted the use
of both DBP and DEHP in pharmaceutical medicines, citing concerns that these
chemicals are associated with health risks. Clearly, phthalates are not
problem-free.
On Wednesday, February 12, Teknor Apex (Pawtucket, RI) will hold a seminar at
MD&M West on the complex issues involved with replacing phthalate plasticizers
in PVC compounds. Led by Galland, the company’s vinyl division industry manager,
the seminar will concentrate on the use of phthalate and nonphthalate
plasticizers in PVC-based and certain non-PVC-based medical devices.
“Phthalate replacement has become important to medical device manufacturers
primarily because environmentally minded NGOs such as Greenpeace waged a
decades-long, unsuccessful campaign against PVC and a more recent successful
effort against phthalates, the primary family of plasticizers used to soften
PVC,” Galland remarks. However, there is scientifically no compelling human
health reason to replace plasticizers like DEHP, Galland adds. And while
researchers have discovered that ingested phthalates, particularly DEHP, can be
causatively linked to liver tumors in rodents, there is no such link in humans
because humans metabolize DEHP and other phthalates differently.
While FDA thinks that the toxic and carcinogenic effects of DEHP demonstrated in
laboratory animals have not been shown in human studies, it acknowledges that
there are certain invasive medical procedures during which exposure to DEHP
could exceed acceptable levels. Thus, FDA focuses on the small subset of medical
devices in which DEHP-containing PVC may come into contact with the tissue of
sensitive patient populations in a manner and for a period of time that may
raise concerns about the aggregate exposure to DEHP. Many devices used in
neonatal intensive care units, according to the agency, meet this criterion.
Affected medical devices could include IV tubing, catheters and cannulae, and
enteral tubing.
Next
|
